Difference between revisions of "Projects:CardiacAblation"
| Line 5: | Line 5: | ||
The high anatomical variability of the heart’s left atrium makes its segmentation a particularly difficult problem. Specifically, the shape of the left atrium cavity, as well as the number and locations of the pulmonary veins connecting to it, vary substantially across subjects. We propose and demonstrate a robust atlas-based method for automatic segmentation of the left atrium in contrast-enhanced magnetic resonance angiography (MRA) images. | The high anatomical variability of the heart’s left atrium makes its segmentation a particularly difficult problem. Specifically, the shape of the left atrium cavity, as well as the number and locations of the pulmonary veins connecting to it, vary substantially across subjects. We propose and demonstrate a robust atlas-based method for automatic segmentation of the left atrium in contrast-enhanced magnetic resonance angiography (MRA) images. | ||
| − | + | == Description == | |
We use a weighted label fusion scheme that assigns higher weights to voxels in training segmentations that are located deeper within the structure of interest and that have similar intensities in training and test images [1]. We also handle varying intensity distributions between images by incorporating iterative intensity equalization in a variant of the demons registration algorithm [13, 14] used for the registration of the training images to the novel test image. We perform the segmentation via a label fusion algorithm [10, 11] that uses a training set of MRA images of different patients with corresponding manual segmentations. We first align the training images to the test subject image to be segmented and apply the resulting deformations to the corresponding manual segmentation label maps to yield a set of left atrium segmentations in the coordinate space of the test subject. These form a non-parametric subject-specific statistical atlas. We then use a weighted voting algorithm to assign every voxel to the left atrium or to the background. | We use a weighted label fusion scheme that assigns higher weights to voxels in training segmentations that are located deeper within the structure of interest and that have similar intensities in training and test images [1]. We also handle varying intensity distributions between images by incorporating iterative intensity equalization in a variant of the demons registration algorithm [13, 14] used for the registration of the training images to the novel test image. We perform the segmentation via a label fusion algorithm [10, 11] that uses a training set of MRA images of different patients with corresponding manual segmentations. We first align the training images to the test subject image to be segmented and apply the resulting deformations to the corresponding manual segmentation label maps to yield a set of left atrium segmentations in the coordinate space of the test subject. These form a non-parametric subject-specific statistical atlas. We then use a weighted voting algorithm to assign every voxel to the left atrium or to the background. | ||
Revision as of 22:02, 30 March 2011
Home < Projects:CardiacAblationBack to NA-MIC Collaborations, MIT Algorithms,
Left atrium segmentation
The high anatomical variability of the heart’s left atrium makes its segmentation a particularly difficult problem. Specifically, the shape of the left atrium cavity, as well as the number and locations of the pulmonary veins connecting to it, vary substantially across subjects. We propose and demonstrate a robust atlas-based method for automatic segmentation of the left atrium in contrast-enhanced magnetic resonance angiography (MRA) images.
Description
We use a weighted label fusion scheme that assigns higher weights to voxels in training segmentations that are located deeper within the structure of interest and that have similar intensities in training and test images [1]. We also handle varying intensity distributions between images by incorporating iterative intensity equalization in a variant of the demons registration algorithm [13, 14] used for the registration of the training images to the novel test image. We perform the segmentation via a label fusion algorithm [10, 11] that uses a training set of MRA images of different patients with corresponding manual segmentations. We first align the training images to the test subject image to be segmented and apply the resulting deformations to the corresponding manual segmentation label maps to yield a set of left atrium segmentations in the coordinate space of the test subject. These form a non-parametric subject-specific statistical atlas. We then use a weighted voting algorithm to assign every voxel to the left atrium or to the background.
Cardiac ablation scar visualization
Atrial fibrillation is one of the most common heart conditions and can have very serious consequences such as stroke and heart failure. A technique called catheter radio-frequency (RF) ablation has recently emerged as a treatment. It involves burning the cardiac tissue that is responsible for the fibrillation. Even though this technique has been shown to work fairly well on atrial fibrillation patients, repeat procedures are often needed to fully correct the condition because surgeons lack the necessary tools to quickly evaluate the success of the procedure.
We propose a method to automatically segment the scar created by RF ablation in delayed enhancement MR images acquired after the procedure. This will provide surgeons with a visualization showing the size, shape and location of the scar, which is information central to evaluating the outcome of the procedure.
Description
We work with two types of images for each patient: MR angiography (MRA) images where the blood pool has a higher intensity than surrounding tissue and post-procedure delayed enhancement MR images (DE-MRI) where a contrast agent has been injected into the patient to enhance the ablation scar. Our approach is to first segment the left atrium in the MRA images using the label fusion algorithm described in [1]. We then transfer this segmentation to the DE-MRI image of the same patient by registering the two images.
Since the ablation scar we are trying to segment is known to be located on the left atrium myocardium, we use this spatial prior information to reduce the search space for the ablation scar to only a small vicinity of the left atrium surface. This avoids many false positives caused by the noise in the DE-MRI images. We will be exploring different segmentation methods in this ongoing work.
Results
Here we present results we have obtained for one subject using our methods. In the following images, we show the left atrium segmentation in the MRA image as an outline in one slice as well as a 3D model.
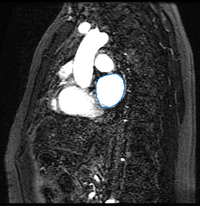
|

|
We then align the MRA and DE-MRI images of the same patient, which allows us to transfer the left atrium segmentation using the resulting deformation field. This is shown in the following figures.
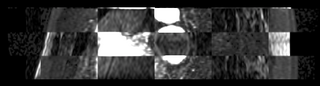
|
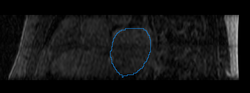
|
Finally, we show some preliminary results of our cardiac ablation scar segmentation obtained using this spatial prior knowledge and intensity thresholding. The figure on the right also shows an expert manual segmentation of the scar alongside our result.
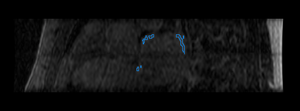
|

|
Literature
[1] Nonparametric Mixture Models for Supervised Image Parcellation, M.R. Sabuncu, B.T.T. Yeo, K. Van Leemput, B. Fischl, and P. Golland. PMMIA Workshop at MICCAI 2009. [2] Diffeomorphic demons: Efficient non-parametric image registration. Vercauteren, T., Pennec, X., Perchant, A., Ayache, N. NeuroImage 45(1), S61–S72 (2009)
Key Investigators
- MIT: Michal Depa and Polina Golland
- BWH: Ehud Schmidt and Ron Kikinis
Publications
NA-MIC Publications Database on Segmentation and Visualization for Cardiac Ablation Procedures