Projects:FieldmapFreeDistortionCorrection
Back to NA-MIC Collaborations, MIT Algorithms
Contents
Fieldmap-Free EPI Distortion Correction
Echo-planar imaging (EPI) is one of the most widely used pulse sequences in functional magnetic resonance imaging (fMRI) due to its high temporal resolution. The ability to acquire an entire volume within seconds makes it a valuable, non-invasive tool for probing dynamic physiological processes such as the blood-oxygenation-level-dependent (BOLD) response. A significant limitation of EPI is its sensitivity to magnetic field inhomogeneity. Perturbations in the field result in signal loss and geometric distortion in EPI data. Previous studies have shown that correcting geometric distortion in functional images increases the accuracy of co-registration to structural MR. Precise anatomical localization of functional activation is especially important in single-subject studies (ie. pre-surgical evaluation) and in cases where the structural MR is used as a reference to sample the functional data (ie. cortical-surface-based analysis). Therefore, field inhomogeneity and distortion is a significant problem in fMRI. In this work, we describe a method for correcting the distortions present in echo planar images (EPI) and registering the EPI to structural MRI. A fieldmap is predicted from a tissue/air segmentation of the MRI using a perturbation method and subsequently used to unwarp the EPI data. Shim and other missing parameters are estimated by registration. We obtain results that are similar to those obtained using fieldmaps, however neither fieldmaps, nor knowledge of shim parameters is required.
Registration without Correction
Localization of functional information relies on accurate registration of EPI and structural MR, which can be difficult due to EPI distortion caused by B0 field inhomogeneity. Correcting distortion using acquired Fieldmaps has been shown to improve registration [1], but Fieldmaps may not be available or may not be applicable if significant motion is present in the EPI, resulting in sub-optimal registration.
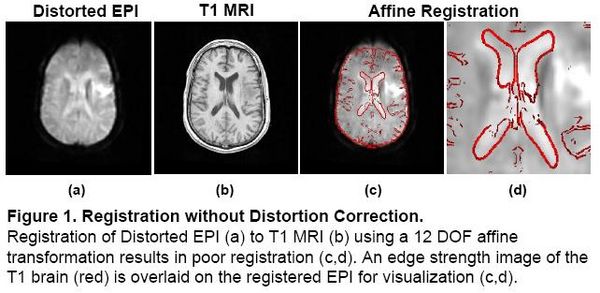
|
Problem 1: Segmentation
Magnetic field models exist to compute a Fieldmap from a Tissue/Air segmentation [2,3], but segmenting structural MR is difficult due to the similar intensities of bone and air. In the Fieldmap-Free method [4], T1 MRI was segmented using a trained classifier that computes the probability of tissue given MR intensity. CT data was used for training and validation only, allowing the trained classifier to be applied to data sets without CT.
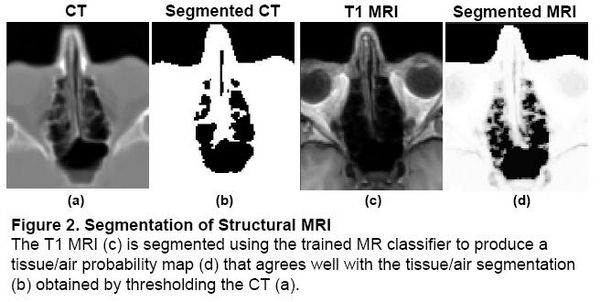
|
Problem 2: Shim Estimation
Existing magnetic field models do not account for the shim fields that reduce B0 field inhomogeniety prior to acquisition. Without this information, accurate unwarping is not possible. In this Method, a Fieldmap (without shim) was computed from the Segmented MR using the field model in [2]. Missing Shim Fields were modeled by spherical harmonic basis functions. Registration was used to search over shim parameters until optimal agreement between the EPI and structural MR was obtained.
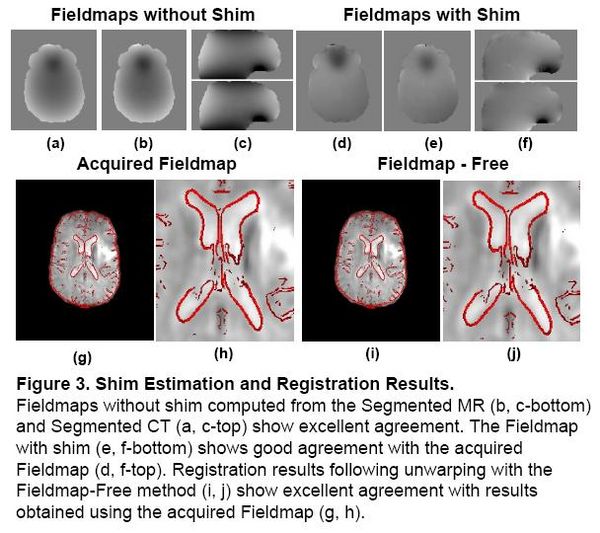
|
Atlas-based Improved Prediction of Magnetic Field Maps
Motivation
In the Fieldmap-Free method described above and in [4], it was shown that tissue/air susceptibility models could be derived from structural MRI by using an intensity-based classifier trained with CT. It was also shown that registration of the EPI and structural MR could be used to search over the unknown shim parameters allowing distortion correction of the EPI that agrees well with results obtained using acquired fieldmaps.
Variability in structural MR acquisitions, however, may limit the efficacy of an intensity-based classifier in cases where the MR intensity properties differ significantly from those of the training data. In [4], CT data sets with MR acquired on the same scanner as the subjects of interest could be used to train the classifier, but this may not be possible in many cases. Limited anatomical information below the brain may also prevent accurate estimation of the perturbing field. Therefore, obtaining more reliable susceptibility models from structural MR is critical for retrospective unwarping of EPI data sets that lack acquired fieldmaps. While previous results predicting fieldmaps from structural MR have shown good agreement with acquired fieldmaps, we hypothesized that improved segmentation methods would result in even greater accuracy.
Description
In this work, anatomical information from a set of 22 whole-head CT data sets is used to achieve improved, subject-specific segmentation of structural MR. A tissue/air atlas is constructed from the CT data to obtain priors on the probability of tissue or air at each location in the anatomy. The corresponding structural MR is used to train a classifier that segments the MR of the subject of interest and this is used as input to a first order perturbation field model to compute a subject-specific fieldmap. The method is evaluated by comparison of predicted fieldmaps and acquired fieldmaps. In addition, the MR classifier can be used to obtain probabilistic bone segmentations from structural MR that show promising agreement with segmented CT.
Results
Atlas Construction
We obtained 22 datasets consisting of CT and MRI from 3 sources: the publicly available Retrospective Image Registration Evaluation (RIRE) database (17 neurosurgery patients), the Radiology department at Brigham and Women's Hospital (BWH) (4 neurosurgery patients) and the Zubal head phantom (1 subject) [5]. For each subject, the CT data was registered to its corresponding MR. The MR was registered to standard space and these transformations were then applied to the co-registered CT. Tissue/air labels were obtained by thresholding the CT data in standard space. Probabilistic tissue/air and air/bone atlases were then constructed as described in [6].
| [[File:Tissue air atlas.jpg | ]] |
[1] Cusack, et al. NeuroImage 18:127-142. 2003.
[2] Jenkinson, et al. Magn Reson Med 52:471-477. 2004.
[3] Koch, et al. Phys Med Biol. 51(24):6381-402. 2006.
[4] Poynton, et al. MICCAI. 271-279, 2008.
[5] Zubal IG, et al. Med. Phys. 21,
299--302. 1994.
[6] Poynton, et al. MICCAI. (in press). 2009.
Key Investigators
- MIT: Clare Poynton, Polina Golland
- BWH/Harvard:Alex Golby, William (Sandy) Wells
- Oxford: Mark Jenkinson
Publications
In Print